Back to Standard Model
Contents
- Fundamental Interactions
- Feynman Diagrams
- Electromagnetic Interaction
- Strong Interaction
- Weak Interaction
Fundamental Interactions
- All forces of nature derive from four Fundamental Interactions (or Forces), which govern how particles interact and, in some cases, decay:
- Electromagnetism
- Strong Force
- Weak Force
- Gravity
- The Standard Model governs the first three; General Relativity the last.
- The forces differ regarding:
- The kinds of particles experiencing the force
- The nature of the particle mediating the force
- The relative strength of the force
- The effective range of the force

Feynman Diagrams
- A Feynman Diagram represents particle interactions.
- The horizontal axis represents space in one dimension, with particles moving left and right along the horizontal line.
- The vertical axis represents time.
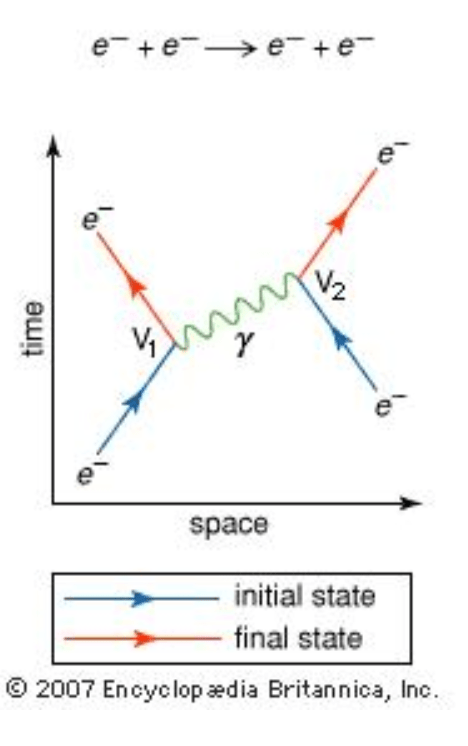
- At right is a Feynman diagram representing the reaction e–+ e– → e–+ e–
- The left electron e− moves to the right (blue line) along the horizontal line.
- The right electron e− then moves to the left (other blue line) along the horizontal line.
- At vertex V1 the left electron emits a photon γ and recoils, moving to the left (red line).
- At vertex V2 the right electron absorbs the photon γ and recoils, moving to the right (red line).
- Lines with arrows, denoting the direction of travel, represent Matter Particles (electrons, neutrinos, quarks, protons, etc)
- Antiparticles have reversed arrows (which doesn’t mean they travel back in time).
- Wavy lines with no arrows represent Exchange Particles (photons, gluons, W and Z bosons).
- A vertex is a junction of three lines.
- Vertices obey the conservation laws for charge, baryon number, and lepton number.
- Only lines entering or leaving the diagram represent observable particles
Electromagnetic Interaction
- The electromagnetic force manifests itself as the attraction and repulsion among electrically-charged particles
- The force is responsible for
- electrons orbiting atomic nuclei
- ionic and covalent bonds
- chemical reactions
- electricity and magnetism
- electromagnetic waves
- Its exchange particle is the photon (γ).
Annihilation
- When a particle and its antiparticle collide, they annihilate each other, releasing energy.
- Positrons (e+) don’t live long because they are attracted to negatively-charged electrons (e‒). When they collide, their masses are converted into energy in the form of a high-energy photon per E=mc2.
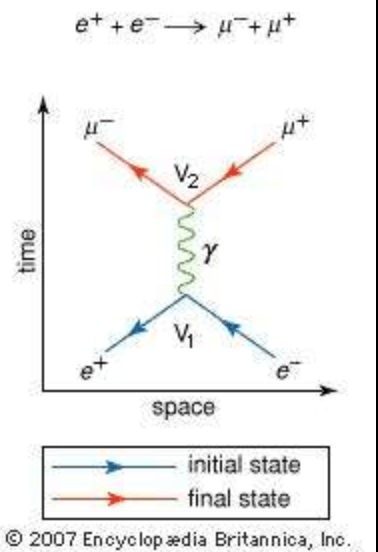
- Positron e+ and electron e‒ approach each other on the horizontal axis, collide, and are annihilated, releasing the photon 𝛾, which splits into a muon (𝜇‒) and an antimuon (𝜇+)
- [Arrows for antimatter particles are always reversed]
- The reaction is: e+ + e‒→ 𝜇‒ + 𝜇+
Pair Production
- Pair production is the creation of a particle and its antiparticle from a high-energy photon. An electron and a positron are created, for example, when a gamma-ray photon passes near the electric field of a large atom such as lead or uranium.
- The energy of the photon is converted into the masses of the electron and positron per E=mc2.
- Any excess energy is converted to kinetic energy of the electron and positron.
- Conserved in the process are electric charge (0), energy, and momentum.
Strong Interaction
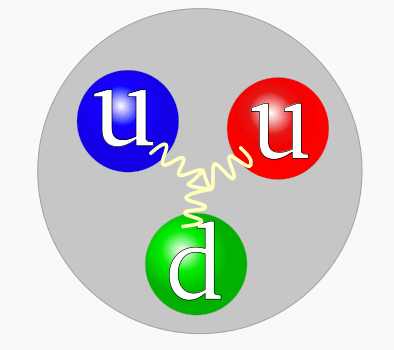
- The Strong Force manifests itself as the attraction and repulsion among color-charged particles.
- Color-Charge
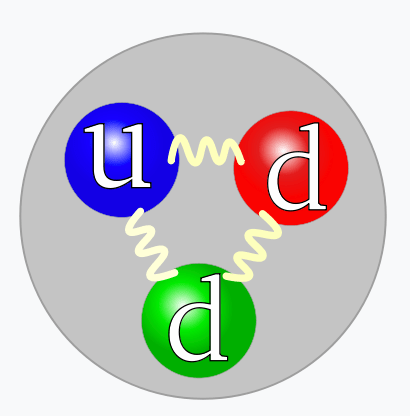
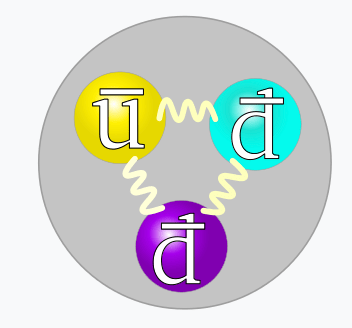
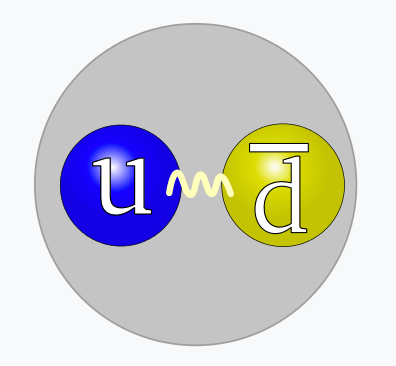
- A quark has a color-charge of red, green, or blue
- An antiquark has a color-charge of antired, antigreen, or antibluel
- Quarks with different color-charges attract
- Quarks with the same color-charge repel
- A hadron is a particle built from quarks:
- A baryon consists of three quarks: red, blue, green.
- An antibaryon consists of three antiquarks: antired, antiblue, antigreen
- A meson (mezz-on) consists of a quark and an antiquark
Baryons
Proton
Neutron
Antibaryons
Antiproton
Antineutron
Mesons
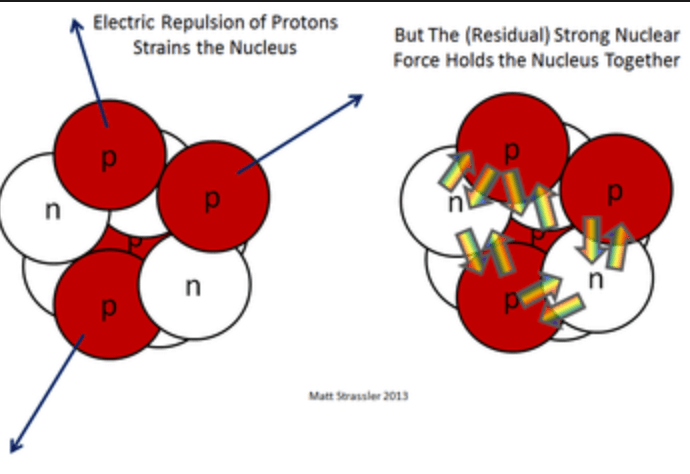
Residual Strong Force
The Residual Strong Force, also known as the Nuclear Force, keeps protons and neutrons together in atomic nuclei, overcoming the electromagnetic repulsion among protons.
Weak Interaction
- The Weak Force manifests itself as a short-range force by which subatomic particles decay or change flavor
- The force is responsible for
- radioactivity
- nuclear fusion
- nuclear fission
- decay of unstable subatomic particles
- Its exchange particles are the W+, W–, and Z0 Bosons
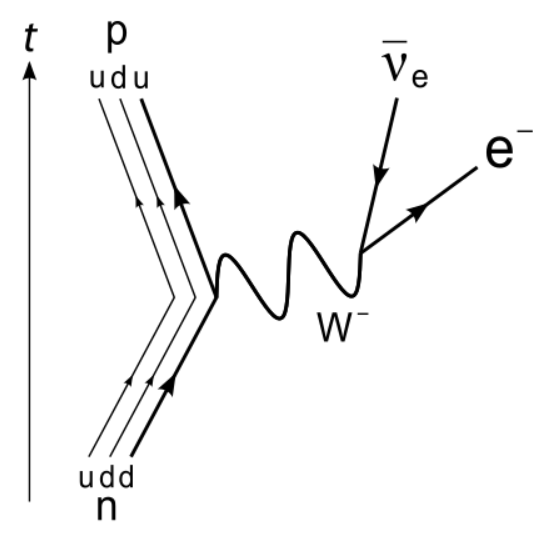
Free Neutron Decay (Electron Emission Beta Decay)
Neutrons are typically bound in the nuclei of atoms, where they are stable. But a neutron on on its own spontaneously decays into a proton, an electron, and an antineutrino, per the weak force. A free neutron, as it’s called, has an average lifetime of only 14 minutes 42 seconds. The type of decay is called Electron Emission Beta Decay. A neutron is made up of two down quarks and an up quark. In the decay, a down quark (d) spontaneously emits a W-, an exchange particle of the weak force, which splits into an electron and an antineutrino.
Carbon Dating (Electron Emission Beta Decay)
- Carbon has three naturally-occurring isotopes
- C-12 makes up 99% of natural carbon
- C-13 makes up about 1%
- C-14, making up a tiny percent, is radioactive, spontaneously decaying into nitrogen.
- Through photosynthesis, plants absorb carbon in the form of carbon dioxide. The carbon is passed onto animals when they eat the plants.
- The ratio of C-14 to C-12 remains constant as long as the organism is alive, since C-14, though decaying, is constantly replenished.
- When the organism dies, however, C-14 is no longer replenished and the ratio of C-14 to C-12 decreases as C-14 atoms decay.
- The ratio of C-14 to C-12 in a fossil can then be used to estimate how long ago the organism died.
- A carbon-14 nucleus, with 6 protons and 8 neutrons, spontaneously decays into nitrogen-14, with 7 protons and 7 neutrons, with a half-life of 5,730 years, emitting an electron and antineutrino in the process. (The mean lifetime of a Carbon-14 atom is 8267 years.)
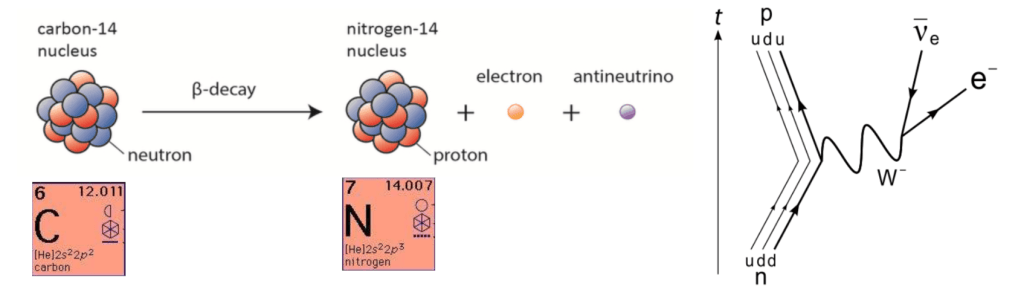
- This happens when a neutron in the carbon-14 nucleus decays into a proton, through Electron Emission Beta Decay. In this process, a down quark (d) changes flavor into an up quark (u), emitting an electron (e–) and an antineutrino (𝜈e).
- Math
- Half-live = (mean lifetime) x Log[2.0]
- λ = decay rate = 1 / (mean lifetime)
- For Carbon-14
- Half-live = (8267 yrs) x Log[2.0] = 5730.25 yrs
- λ = decay rate = 1 / 8267 = 0.000120963 yrs-1
- Probability that the lifetime of a particular Carbon-14 is longer than 10,000 years = 0.30
- N[Probability[t > 10000,t ⩬ ExponentialDistribution[1/8267]]] = 0.298308
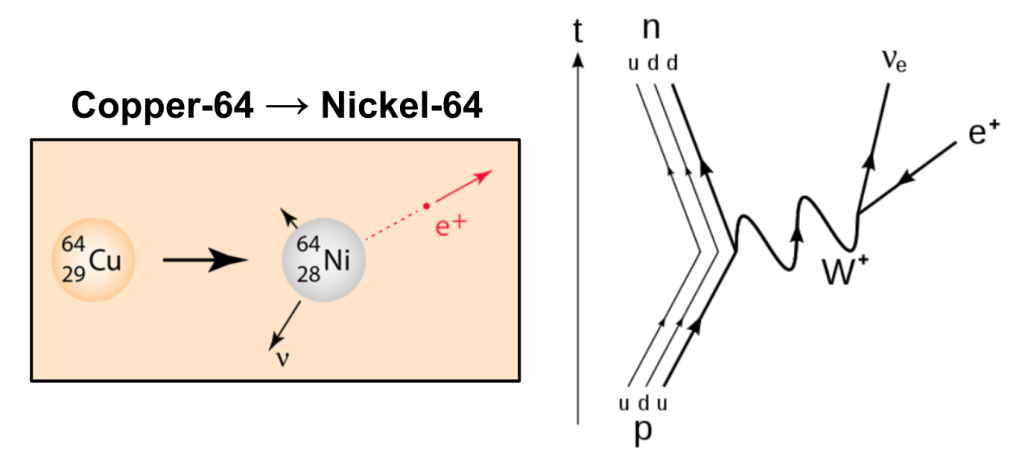
Radioactivity (Positron Emission Beta Decay)
In Positron Emission Beta Decay an up quark in a proton changes flavor into a down quark, changing the proton to a neutron and emitting a neutrino (𝜈e) and a positron (e+). The process thus produces a daughter nucleus with an atomic number one less than its parent but with the same mass number. Positron emission was first observed by Irène and Frédéric Joliot-Curie in 1934.
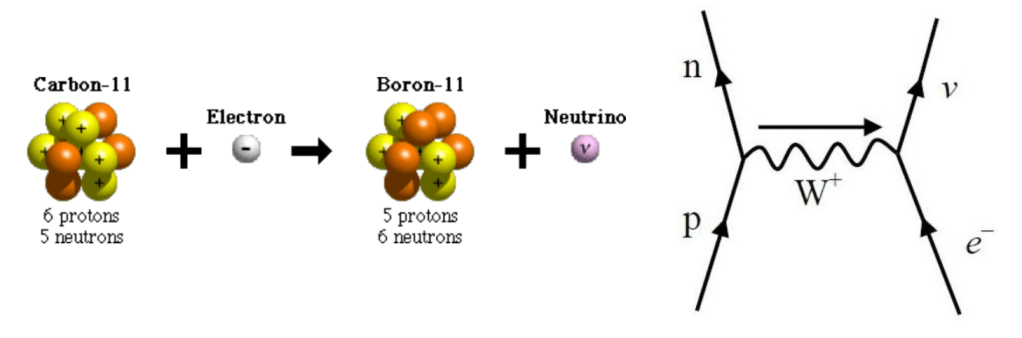
Radioactivity (Electron Capture Beta Decay)
In Electron Capture Beta Decay an orbital electron in an atom’s inner shell is captured by the nucleus where it combines with a proton, which decays into a neutron and emits a neutrino. The atom is therefore lowered by one atomic number, e.g. from Carbon with 6 protons to Boron with 5