Back to Energy
Contents
- Work
- Forms of Energy
- The Two Basic Forms of Energy
- Potential and Kinetic Energy of the Pendulum
- Work and Heat: Forms of Energy Transfer
- Law of Conservation of Energy, Emmy Noether’s Proof
- Conservation of Energy in Classical and Quantum Mechanics
- Kinetic Energy in Classical and Relativistic Mechanics
Work
- Energy is the capacity for doing work.
- Work is the energy transferred as a force moves an object
- britannica.com/science/work-physics
- “Work is the measure of energy transfer that occurs when an object is moved over a distance by an external force. If the force is constant, work may be computed by multiplying the length of the path by the magnitude of the force.”
- “Work done on a body is equal to the increase in the energy of the body, for work transfers energy to the body.}
- “Work and energy are expressed in the same units, for example, joules and foot-pounds.”
- Formal Definition
- Work by a force on an object over a distance is the force exerted times the distance covered.
- For a constant force in a straight line
- W = F x D
- For a variable force in a straight line from a to b
- Dropping a bowling ball
- If a sixteen pound bowling ball is dropped five feet, Earth’s gravity does work on the ball equalling:
- 16 pounds of force x 5 feet travelled = 80 foot-pounds of work.
- If a sixteen pound bowling ball is dropped five feet, Earth’s gravity does work on the ball equalling:
- The cue ball, with 10.6 joules of kinetic energy, hits a stationary eight ball:
- Before impact:
- Cue ball has kinetic energy of 10.6 joules
- Eight ball has zero.
- At Impact:
- 10.6 joules of work is performed, transferring 10.6 joules of energy from the cue ball to the eight ball
- After impact:
- The cue ball has zero kinetic energy
- Eight ball has kinetic energy of 10.6 joules
- Before impact:
- The formula W = F x D is used to derive formulas for different forms of energy
- To derive the formula for kinetic energy, for example, you calculate the amount of work required to move a mass m from rest to v meters per second.
- That amount of work is ½ mv2 joules.
Forms of Energy
The Forms
- Total energy
- The total energy of a system is the sum of the forms of energy in the system, each calculated by its formula.
- Forms of energy include
- Chemical energy
- Elastic potential energy
- Electrical energy
- Gravitational potential energy
- Kinetic energy
- Mass energy
- Nuclear energy
- Radiant energy
- Thermal energy
Their Formulas

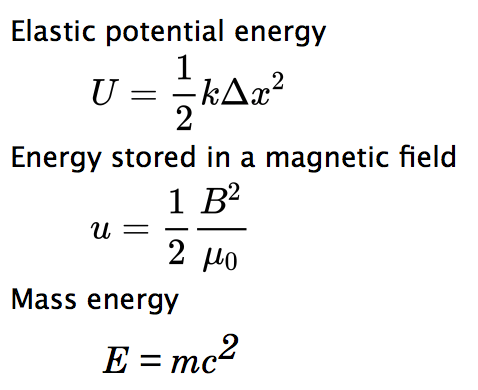
The Two Basic Forms of Energy
- Kinetic Energy, the energy of motion
- The kinetic energy of an object moving at a given speed is the work required to accelerate the object from rest to that speed.
- KE = ½ mv2 is derived from W = FD and F = MA
- Potential Energy, the energy of location (in a force field)
- The potential energy of an object at a given location is the work required to move the object from a reference point to that location.
- For example, the formula for gravitational potential energy,
- is derived from W = FD and Newton’s Law of Universal Gravitation.
- Force and potential energy are intimately connected:
- Force = change of potential energy divided by distance
- For example, electric forces among atoms bend water molecules
- In the diagram the forces push downhill, from higher potential energy to lower.
- The potential energy of an object at a given location is the work required to move the object from a reference point to that location.
Potential and Kinetic Energy of the Pendulum
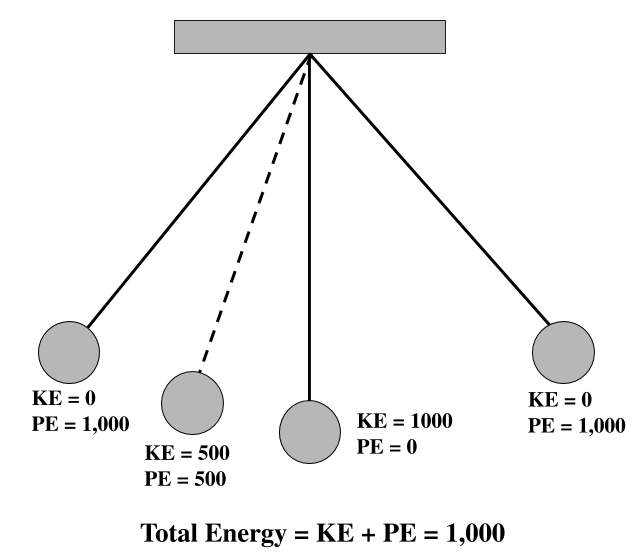

- The total energy of a pendulum system at a time = the kinetic energy of the bob at the time + the gravitational potential energy of the system at the time.
- The kinetic energy of the bob, calculated by ½ mv2, changes from zero to the total energy as the bob goes from its high point to its low point
- m = mass of bob
- v = velocity of bob
- The gravitational potential energy, calculated by gmh, changes from the total energy to zero as the bob goes from its high point to its low point
- g = acceleration due to gravity
- m = mass of bob
- h = height of bob
Work and Heat: Forms of Energy Transfer
- Work is a form of energy transfer, not a form of energy
- Work is the energy transferred resulting from a force moving an object
- Heat is a form of energy transfer, not a form of energy
- Heat is the energy transferred resulting from a difference in temperature
- The total energy of a system does not include work and heat.
- Water Bucket Analogy
- A first bucket has five quarts of water and a second two. You pour one quart from the first bucket into the second. The total amount of water, seven quarts, remains the same but it’s now divided differently: 4 + 3 vs 5 + 2.
- The seven quarts does not include the one quart transferred.

Law of Conservation of Energy,
Emmy Noether’s Proof
- In 1915 Emmy Noether (EM-mee ΝEW-tar) proved Noether’s Theorem, which says:
- If the laws of physics remain valid through transformation X, then quantity Y is conserved, where
- X = translations in time and Y = energy
- X = translations in space and Y = linear momentum
- X = rotations in space and Y = angular momentum
- X = gauge transformations and Y = electric charge
- If the laws of physics remain valid through transformation X, then quantity Y is conserved, where
- Thus, Noether proved that the Law of the Conservation of Energy follows from the fact that the laws of physics remain valid through time.

Conservation of Energy in Classical and Quantum Mechanics
- Proof of Conservation of Energy in Classical Mechanics
- E = ½ mv2 + V(x)
- The total energy of a system = kinetic energy + potential energy
- [several steps of reasoning]
- Therefore, dE/dt = 0
- The rate of change of the total energy of the system = 0
- E = ½ mv2 + V(x)
- Proof of Conservation of Energy in Quantum Mechanics
- For any operator Q, d/dt ⟨Q⟩ = -i/ℏ ⟨[Q,H]⟩, where H represents the total energy of a system
- The time rate of change of the expectation of an operator Q equals -i/ℏ times the expectation of the commutator of Q with H
- [H,H] = 0
- Every operator commutes with itself
- Therefore, d/dt ⟨H⟩ = -i/ℏ ⟨[H,H]⟩ = 0
- The rate of change of the expectation of the Hamiltonian = 0
- For any operator Q, d/dt ⟨Q⟩ = -i/ℏ ⟨[Q,H]⟩, where H represents the total energy of a system
Kinetic Energy in Classical and Relativistic Mechanics
Derivation of Kinetic Energy from Momentum
- KE = ∫ F dx
- KE = the kinetic energy of a particle = the quantity of work required to increase the velocity of the particle from zero to v
- Work = force x distance (F dx)
- ∫ F dx = the integral of F dx, that is, (very) roughly, the sum of the force F times each tiny piece of distance.
- KE = ∫ dp/dt dx
- F = dp/dt, i.e. force equals the time rate of change of momentum.
- So 2 follows from 1 by replacing F with dp/dt.
- KE = ∫ dp/dt v dt
- From 2 since dx = v dt, distance = velocity times time.
- KE = ∫ dp/dv dv/dt v dt
- From 3: differential chain rule: dp/dt = dp/dv dv/dt
- KE = ∫ dp/dv a v dt
- From 4: acceleration = time rate of change of velocity: dv/dt = a
- KE = ∫ dp/dv v dv
- From 5: velocity = acceleration times time: a dt = dv
- Continued below
Continuation: Kinetic Energy in Classical Mechanics
- 7. KE = ∫ d(mv)/dv v dv
- From 6: momentum = mass times velocity: p = mv
- 8. KE = 1/2 mv2
- Mathematica
- Integrate[D[m v, v] v, v] = mv2/2
- Mathematica
Continuation: Kinetic Energy in Relativistic Mechanics
- 7. KE = ∫ d(mvγ)/dv v dv
- From 6: momentum = mass times velocity times the Lorentz Factor : p = mvγ
- where the Lorentz Factor γ = 1/√(1 – v2/c2)
- From 6: momentum = mass times velocity times the Lorentz Factor : p = mvγ
- 8. KE = γmc2 = mc2/√(1 – v2/c2)
- Mathematica
- FullSimplify[Integrate[D[γ m v, v] v, v]]
- = mc2/Sqrt[1 – v2/c2]
- FullSimplify[Integrate[D[γ m v, v] v, v]]
- Mathematica
E = mc2
- E = mc2 says that the energy of a particle at rest equals its mass times the speed of light squared.
- The equation follows directly from the relativistic kinetic energy of particle, γmc2.
- γmc2 = mc2/√(1 – v2/c2)
- v = 0
- Therefore, γmc2 = mc2/√(1 – 0/c2) = mc2
- That is, mc2 is the “kinetic” energy of a particle at rest.
E = mc2 and Approximate Kinetic Energy
- A useful approximation of 1/√(1 – x2), where x is small, is 1 + x2/2.
- For example, where x = 1/1000:
- 1/√(1 – x2) = 1.000000500000375000312500
- 1 + x2/2 = 1.000000500000000000000000
- For example, where x = 1/1000:
- KE = mc2/√(1 – v2/c2)
- Therefore, KE ≅ mc2 (1 + (v/c)2/2) = mc2 + 1/2 mv2
- Mathematica
- Expand[m c^2 (1 + (v/c)^2/2)]
- = c^2 m + (m v^2)/2
- Expand[m c^2 (1 + (v/c)^2/2)]
- Mathematica
- Thus, the energy of a particle is its rest energy plus its approximate kinetic energy in Classical Mechanics.
E = mc2 in Action: Particle Annihilation
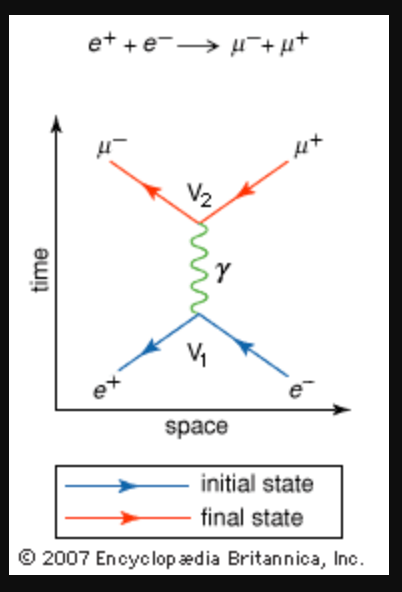
- In the Feynman Diagram at right an electron (e–) and its antiparticle, the positron (e+), annihilate each other when they collide at V1, creating a gamma ray photon γ, which at V2 becomes a muon (𝜇–) and an antimuon (𝜇+). Energy is conserved throughout the process:
- me+c2/√(1 – v2/c2) + me-c2/√(1 – v2/c2) = the energy of the gamma ray photon = mμ+c2/√(1 – v2/c2) + mμ–c2/√(1 – v2/c2).