Contents
- Thermodynamics
- Zeroth Law of Thermodynamics
- First Law of Thermodynamics
- Second Law of Thermodynamics
- Third Law of Thermodynamics
Thermodynamics
- Thermodynamics is the science of energy, temperature, heat, and work.
- Energy is an abstract quantity, the capacity for doing work, that retains the same value through changes of its forms.
- Basic Forms:
- Kinetic Energy, the energy of motion
- The kinetic energy of an object moving at a given speed is the work required to accelerate the object from rest to that speed.
- Potential Energy, the energy of location (in a force field)
- The potential energy of an object at a given location is the work required to move the object from a reference point to that location.
- Kinetic Energy, the energy of motion
- Basic Forms:
- The temperature of a physical system is the average kinetic energy of its constituent particles.
- Heat is energy transferred as the result of a difference in temperature.
- Work is energy transferred by a force moving an object.
- Work and heat are forms of energy transfer, not forms of energy like kinetic and potential energies.
- The total energy of a system includes kinetic and potential energies but not work and heat.
- Energy is an abstract quantity, the capacity for doing work, that retains the same value through changes of its forms.
View Energy
Zeroth Law of Thermodynamics
- Zeroth Law
- If two systems are in thermal equilibrium with a third, they are in thermal equilibrium with each other.
- Systems are in thermal equilibrium if they are the same temperature
- A system in thermodynamic equilibrium has no tendency to spontaneously change.
First Law of Thermodynamics
Concepts
- The internal energy of a system is the total kinetic and potential energies of its constituent particles.
- Internal energy does not include the kinetic and potential energies of the system as a whole. The kinetic energy of a test tube thrown across the room is not part of the internal energy of its contents.
- Heat is the energy transferred from one body to another as the result of a difference in temperature.
- Work is the energy transferred as the result of a force moving an object.
First Law
- The change in a system’s internal energy equals the heat added to the system from its surroundings minus the work done by the system on its surroundings.
- That is, change in internal energy ΔU = heat added Q – work done W
- ΔU = Q – W

Part (a): heat Qin is added to the system (cylinder + piston), a force F=PA is exerted on the piston, and ΔUa = Qin.
Part (b): the force does work Fd = Wout on the piston and ΔUb = -Wout
Therefore: ΔU = ΔUa + ΔUb = Qin – Wout
Derivation of First Law from the Law of Conservation of Energy
- Law of Conservation of Energy
- The total energy of an isolated system remains the same.
- An isolated system exchanges no energy or matter with its surroundings.
- The total energy of an isolated system remains the same.
- We want to derive the First Law of Thermodynamics
- The change in a system’s internal energy equals the heat added to the system from its surroundings minus the work done by the system on its surroundings.
- Let X be a system S plus its surroundings R, the so-called Universe.
- X is an isolated system.
- Additional assumption #1
- So, the total energy of X remains the same.
- Therefore any change in S’s energy is offset by a change in R’s energy.
- S and R exchange energy only by work and transfer of heat.
- Additional assumption #2
- Therefore S changes energy only by exchanging work heat with R.
- S’s only energy is internal energy
- Additional assumption #3
- Therefore, S’s internal energy changes only by exchanging work and heat with R.
Second Law of Thermodynamics
Second Law
- The entropy of an isolated system either increases or stays the same.
Isolated System
- An isolated system exchanges no energy or matter with its surroundings.
Entropy
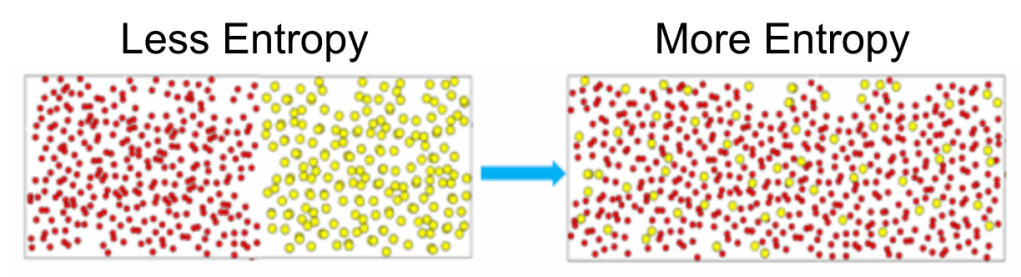
- The system on the right is less ordered, more random, its energy more dispersed
- Entropy is the measure of such disorder, randomness, and dispersion of energy.
Clausius Definition of Change of Entropy (Classical Thermodynamics)

The increase in the entropy ΔS of a system equals the amount of heat added Q divided by its absolute temperature T.
Example
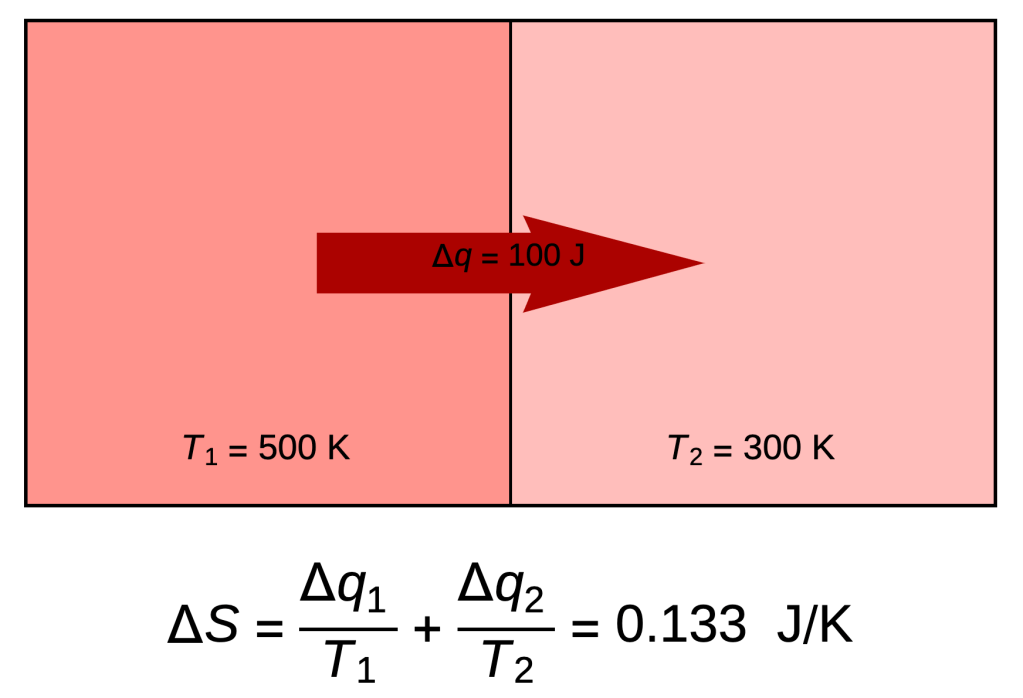
- The system on the right consists of two compartments of carbon dioxide: one at 500 Kelvin (440.33 Fahrenheit), the other at 300 Kelvin (80.33 Fahrenheit).
- A divider between the compartments is removed and 100 Joules of heat flows from left to right.
- As the molecules mix and mingle, the system undergoes an increase of entropy ΔS = -100/500 + 100/300 = 0.133 Joules / Kelvin.
View Interactive Entropy, Temperature, and Heat
Boltzmann Definition of Entropy (Statistical Thermodynamics)

- The entropy S of an isolated system equals k ln Ω
- where k is the Boltzmann Constant, 1.380649 x 10-23 J/K (Joules per Kelvin)
- ln is the natural logarithm
- Ω is the number of microstates consistent with a given macrostate, e.g. temperature, pressure, volume
Artificial Example
- Consider a system of 25 circles, green or blue. The macrostates of the system are the number of blue circles, from 0 to 25 (total 26). The microstates are the color combinations of the circles, from 0 to 33554431 (total 33554432).
- Here are three microstates for macrostate #12:



- There are 9.6577 x 106 microstates for macrostate #12.
- So the entropy of the #12 system is k ln 9.6577 x 106 = 16.0833 k
- Here are three microstates for macrostate #2:



- There are 325 microstates for macrostate #2.
- So the entropy of the #2 system is k ln 325 = 5.78383 k
The entropy of the 12-blue system (16.0833 k ) is thus greater than that of the 2-blue system (5.78383 k).
View Interactive Macrostates and Microstates
Probabilistic Nature of the Second Law
Molecules in Two Compartments
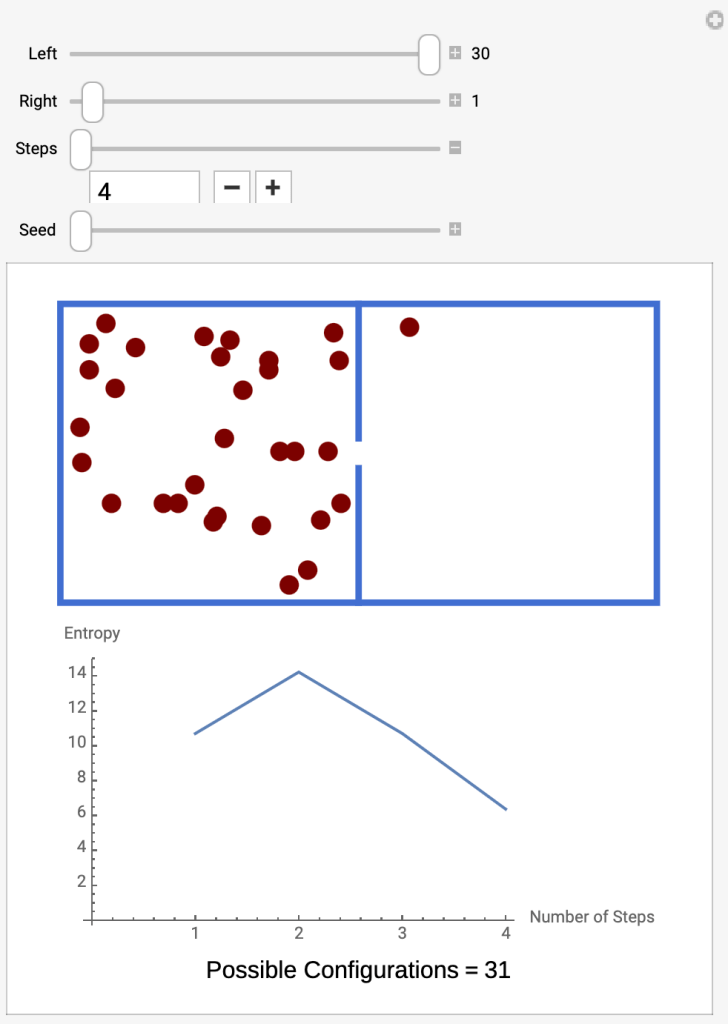
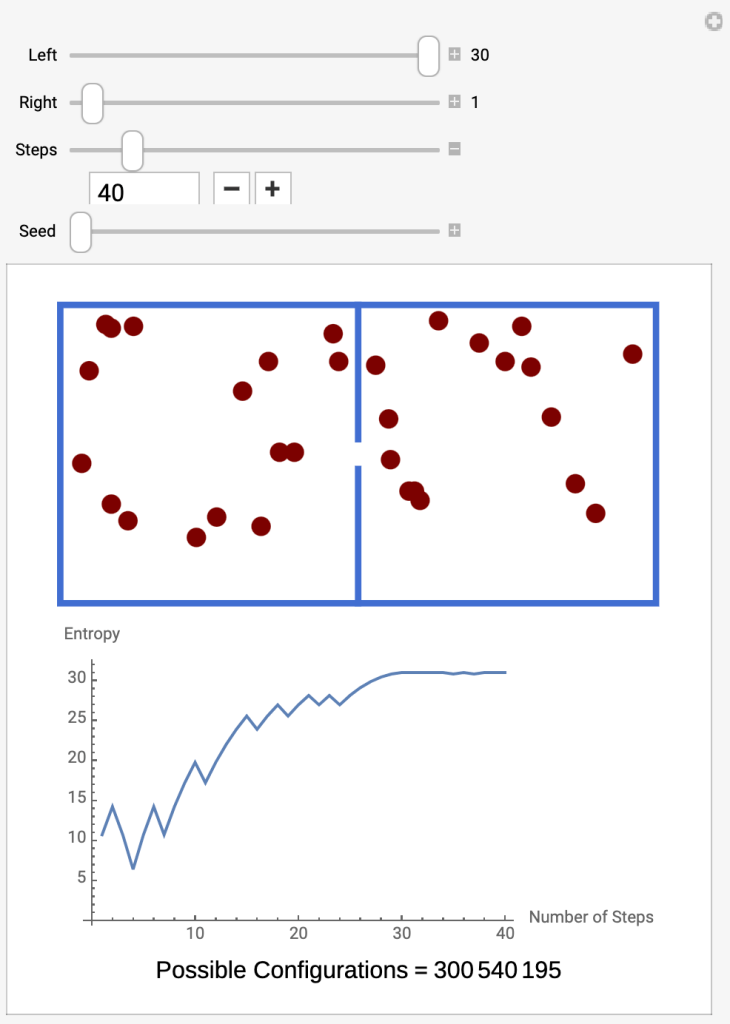
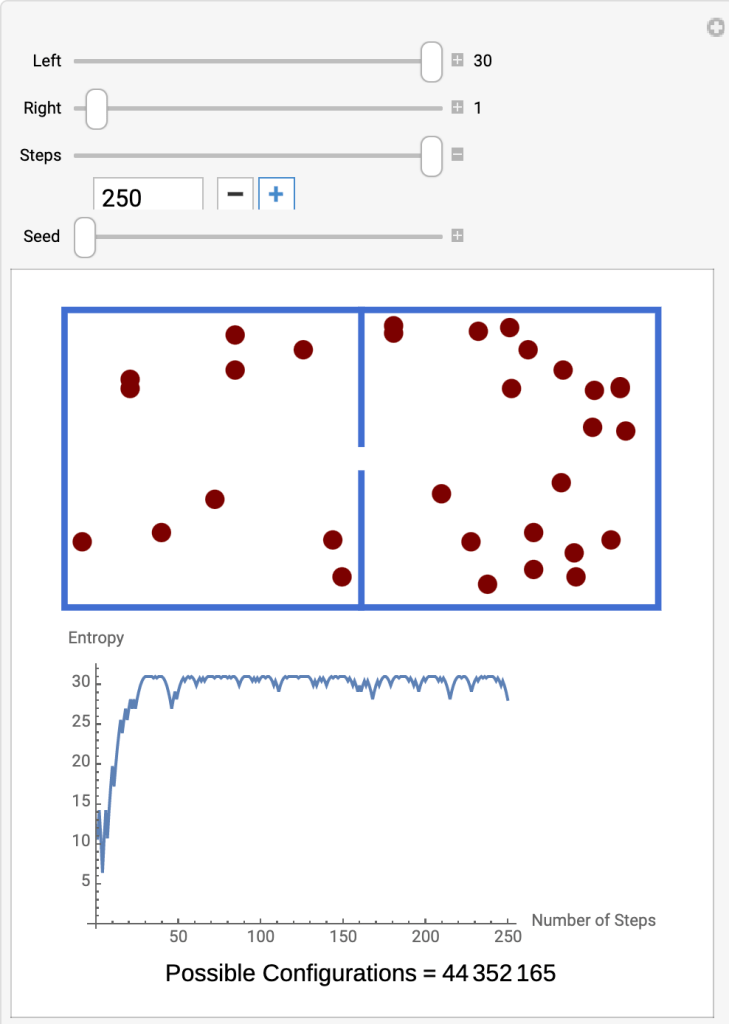
- Run the Interactive Molecules in Two Compartments
- At the beginning (Step 4) there are 30 molecules in the left compartment and 1 in the right. The graph shows the entropy at 6.
- Click the plus sign under Steps to advance to the next step.
- Each step moves one particle through the gap, the probability of its direction determined by the number of molecules on each side. The more molecules on a side, the greater the probability that a molecule from that side will pass through the gap.
- By Step 40 the entropy has increased to 30, attaining thermodynamic equilibrium.
View Interactive Molecules in Two Compartments
Chemical Reactions
- britannica.com/science/chemical-reaction
- “A chemical reaction will favor the formation of products if the sum of the changes in entropy for the reaction system and its surroundings is positive.”
- “An example is burning wood. Wood has a low entropy. When wood burns, it produces ash as well as the high-entropy substances carbon dioxide gas and water vapor. The entropy of the reacting system increases during combustion. Just as important, the heat energy transferred by the combustion to its surroundings increases the entropy in the surroundings. The total of entropy changes for the substances in the reaction and the surroundings is positive, and the reaction is product-favored.”
Heat Death of the Universe
- britannica.com/science/thermodynamics
- “If the universe is an isolated system, then its entropy must increase with time. Indeed, the implication is that the universe must ultimately suffer a “heat death” as its entropy progressively increases toward a maximum value and all parts come into thermal equilibrium at a uniform temperature. After that point, no further changes involving the conversion of heat into work would be possible. In general, the equilibrium state for an isolated system is precisely that state of maximum entropy.”
Third Law of Thermodynamics
- The entropy of an isolated system nears zero as its temperature approaches absolute zero.
- Per Boltzmann’s equation, the entropy of a system with one microstate is zero:
- S = k ln Ω = k ln 1 = 0