Back to Scientific Theories
Contents
- Chemistry
- Theory of Chemistry
- Deriving Predictions
- Quantum Atomic Model
- Periodic Table and Trends
- Chemical Bonds
- Chemical Reactions
- Combustion of Methane
- Photosynthesis
- Carbonation
- Addendum
Chemistry
- Chemistry is the branch of science dealing with
- the composition, structure, and properties of chemical substances
- the transformation of chemical substances into different chemical substances.
- Terms
- A chemical substance is an element or chemical compound.
- An element consists of only one kind of atom.
- Hydrogen, oxygen, carbon, gold, iron, aluminum.
- A chemical compound consists of one type of molecule, ionic structure, or metallic crystal, each type composed of at least two kinds of atoms.
- Carbon dioxide, water, sodium chloride (salt), amino acids, proteins, DNA
- A mixture consists of more than one type of molecule, ionic structure, or metallic crystal.
- Air, seawater, alloys
- Branches
- Physical Chemistry
- Deals with the principles of chemistry
- Organic Chemistry
- Deals with carbon and its compounds
- Inorganic Chemistry
- Deals with elements other than carbon and its compounds
- Physical Chemistry
Theory of Chemistry
Big Picture

What’s it Designed to Explain
- The composition, structure, and properties of chemical substances
- The transformation of chemical substances into different chemical substances
Postulate of Chemistry
- Chemical substances consist of atoms, nuclei, electrons, ions, and molecules whose structure and interactions are governed by
- the electrostatic force per Quantum Mechanics
- the Laws of Thermodynamics
- the Conservation of Mass for chemical reactions
Particles of Chemistry
- Atoms
- Atomic Nuclei (Protons + Neutrons)
- Bound Electrons
- Ions
- A positive ion, or cation, is an atom with a positive electric charge, having lost an electron.
- A negative ion, or anion, is an atom with a negative electric charge, having acquired an electron.
- Molecules
- A particle consisting of more than one atom
- Free Electrons
Electrostatic Force
- The basic idea of the electrostatic force is that:
- Like electric charges repel.
- Unlike charges attract.
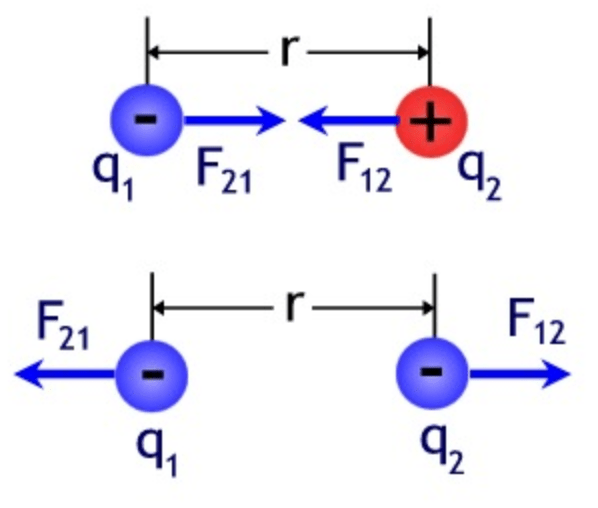
- Coulomb’s Law governs the electrostatic force in classical physics:
- For any pair of charged particles there’s a force F on each, toward the other, with magnitude kq1q2/r2
- where k equals Coulomb’s constant 8.99 × 109
- q1 and q2 are the electric charges of the particles
- r is the distance between them
- For any pair of charged particles there’s a force F on each, toward the other, with magnitude kq1q2/r2
- That is:
- F = kq1q2/r2
- In Quantum Mechanics the electrostatic force is expressed as the electric potential energy of a physical system.
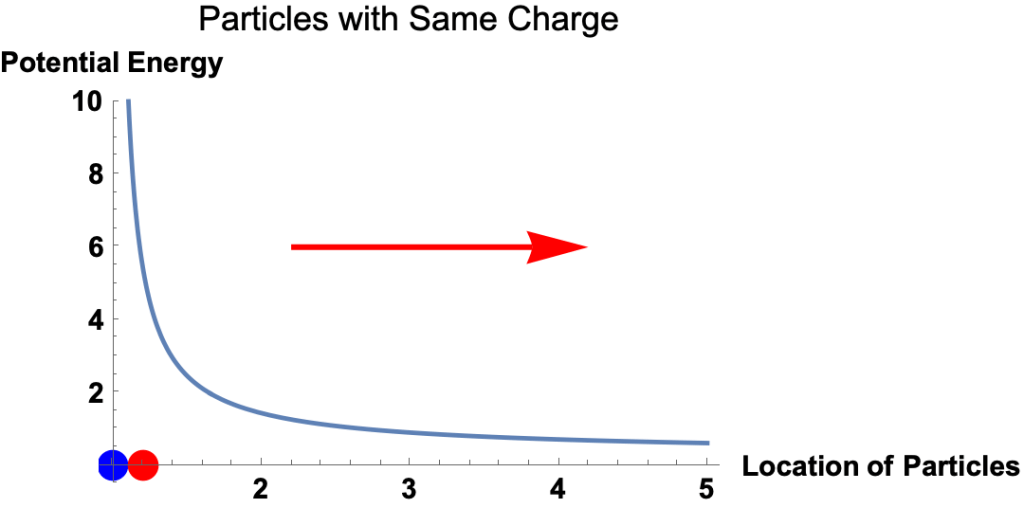
Electric Potential Energy
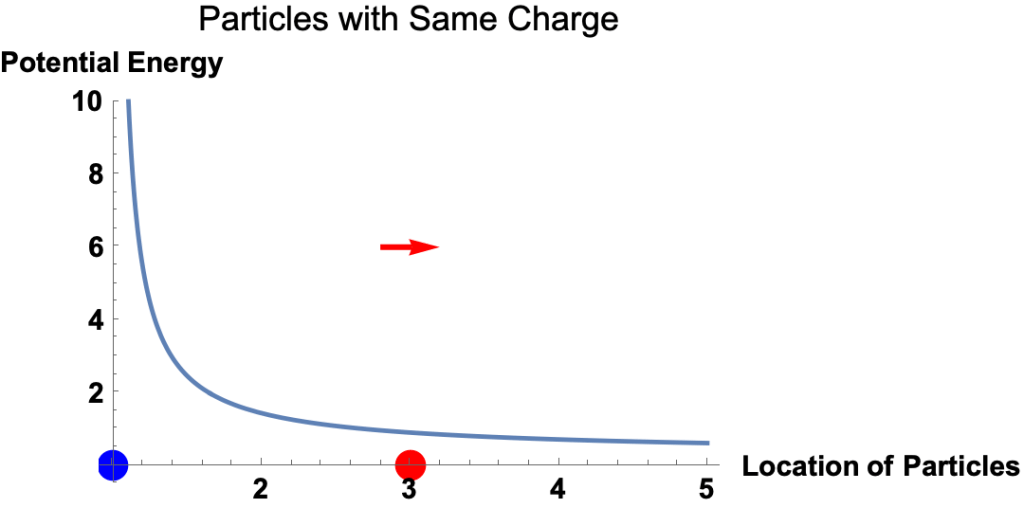
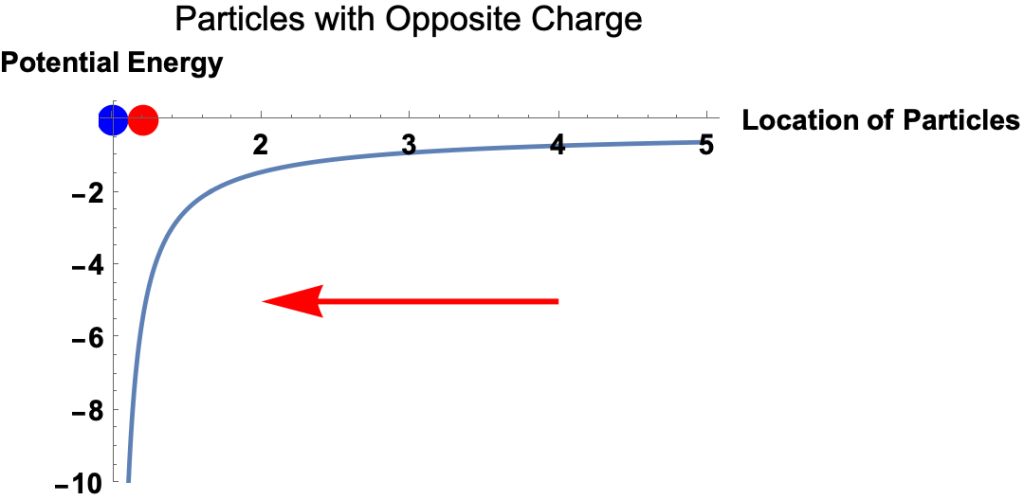
- Potential energy is the energy a particle has because of its location in a force field. It’s the amount of work required to move the particle from a reference point to its current location.
- The electrostatic force on a charged particle at a given location equals the slope of the electric potential energy at the location.
- That is
- The magnitude of the force equals the magnitude of the slope.
- The direction of the force is
- to the right if the potential energy curve is downward to the right
- to the left if the potential energy curve is upward to the right,
Example: Red and Blue Particles with the Same Charge
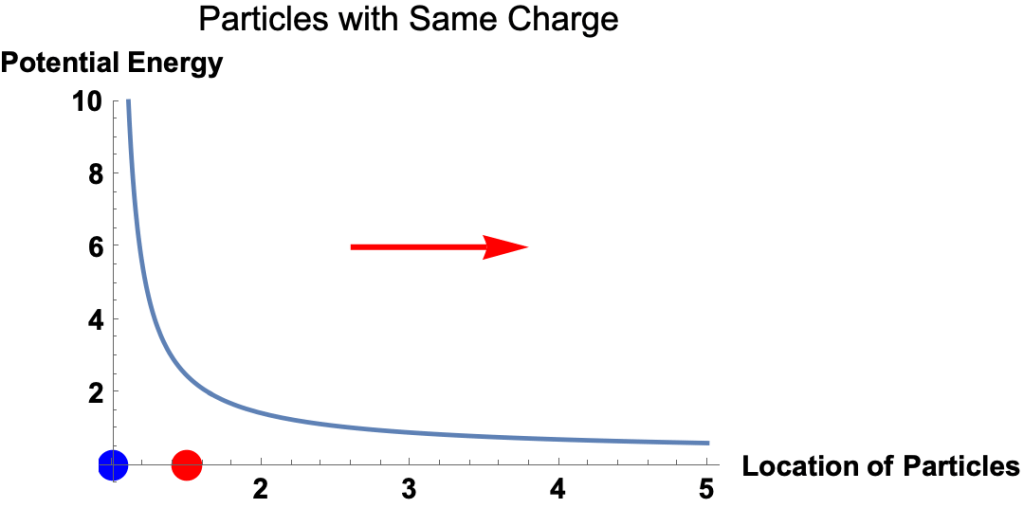
Example: Red and Blue Particles with Opposite Charges
Quantum Mechanics
- Quantum Mechanics is the theory of the interactions among atoms, subatomic particles, and electromagnetic radiation.
- In Classical Mechanics, Newton’s Equation of Motion, F=MA, translates Coulomb’s Law into predictions about a charged particle’s motion.
- In Quantum Mechanics, the Schrödinger Equation, H|Ψ⟩ = E|Ψ⟩, translates electric potential energy into predictions about the movement of a charged particle. The predictions are very different from those Classical Mechanics.
View Quantum Mechanics
Thermodynamics
- Thermodynamics is the science dealing with the relations among heat, work, temperature, and energy.
- Heat is energy transferred as the result of a difference in temperature.
- The temperature of a physical system is the average kinetic energy of its constituent particles.
- Work is energy transferred by a force moving an object
- Energy is the capacity to do work
View Thermodynamics
Conservation of Mass for Chemical Reactions
- The total mass of a system undergoing a chemical reaction remains the same.
- The restriction to chemical reactions is necessary since mass can be converted to energy in nuclear reactions per E=mc2
- Example:
- CH4(g) + 2O2(g) → CO2(g) + 2H2O(g)
- Reactant Side
- Mass of CH4 = 16.043 g/mol
- Mass of O2 = 31.998 g/mol
- Total = 16.043 + 2(31.998) = 80.039 g/mol
- Product Side
- Mass of CO2 = 44.009 g/mol
- Mass of H2O = 18.015 g/mol
- Total = 44.009 + 2(18.015) = 80.039 g/mol
Predictions
- Structure of Atoms
- Periodic Table and Trends
- Nature of Chemical Bonds
- Nature of Chemical Reactions
Deriving Predictions
Complexities
- Solving the equations of Quantum Mechanics precisely is often impossible.
- britannica.com/science/quantum-mechanics-physics/Schrodingers-wave-mechanics
- “The Schrödinger equation cannot be solved precisely for atoms with more than one electron. The principles of the calculation are well understood, but the problems are complicated by the number of particles and the variety of forces involved. The forces include the electrostatic forces between the nucleus and the electrons and between the electrons themselves, as well as weaker magnetic forces arising from the spin and orbital motions of the electrons.”
- britannica.com/science/quantum-mechanics-physics/Schrodingers-wave-mechanics
- Quantum Mechanics predicts probabilities rather than certainties.
- Atoms range from hydrogen, with one proton and one electron, to Oganesson, with 118 protons and 118 electrons.
- Atoms form complicated molecules
- Matter consists of astronomically large numbers of particles
- Matter has different states
Approximate Methods and Models
- To derive predictions chemists use methods and models that simplify matters at the cost of precision. For example:
- Hartree-Fock Method, or variant, for deriving results from Quantum Mechanics
- Born-Oppenheimer Approximation, regarding atomic nuclei as stationary
- Quantum Atomic Model and Orbitals
- Molecular Orbital Theory
- Ball and Stick Diagrams
- Structural Formulas
- A model is a simple representation of complex phenomena.
- Scientific models are used for visualizing, explaining, and forecasting.
View Scientific Models
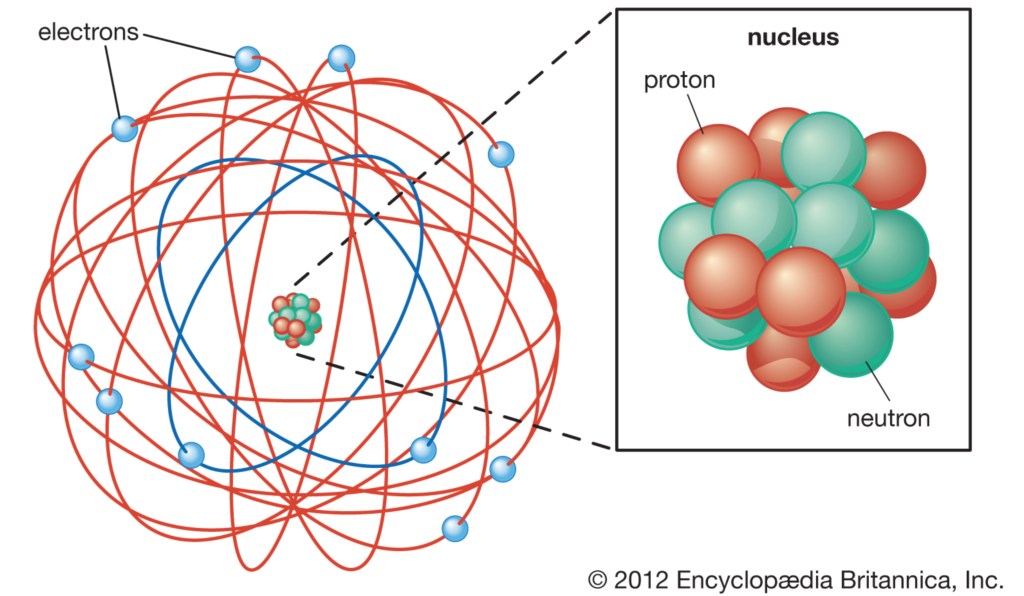
Quantum Atomic Model
- Rutherford Atomic Model of 1911
- Quantum Atomic Model
- 1s, 2s, 2px, 2py, and 2pz Orbitals
- How electrons of first ten elements fill orbitals
- Hartree-Fock Method and Radial Probability Density
Rutherford Atomic Model of 1911
- Ernest Rutherford’s idea was that electrons orbit an atomic nucleus like planets orbit the Sun. Newton’s Law of Universal Gravitation governs the orbits of the planets:
- F = Gm1m2/r2
- Coulomb’s Law was supposed to govern the orbits of electrons:
- F = Kq1q2/r2
- But the Rutherford Atomic Model failed. The problem was not Coulomb’s Law, but Newton’s Equation of Motion, F=MA.
- The solution was to replace Classical Mechanics with Quantum Mechanics and F=MA with the Schrödinger Equation.
Quantum Atomic Model
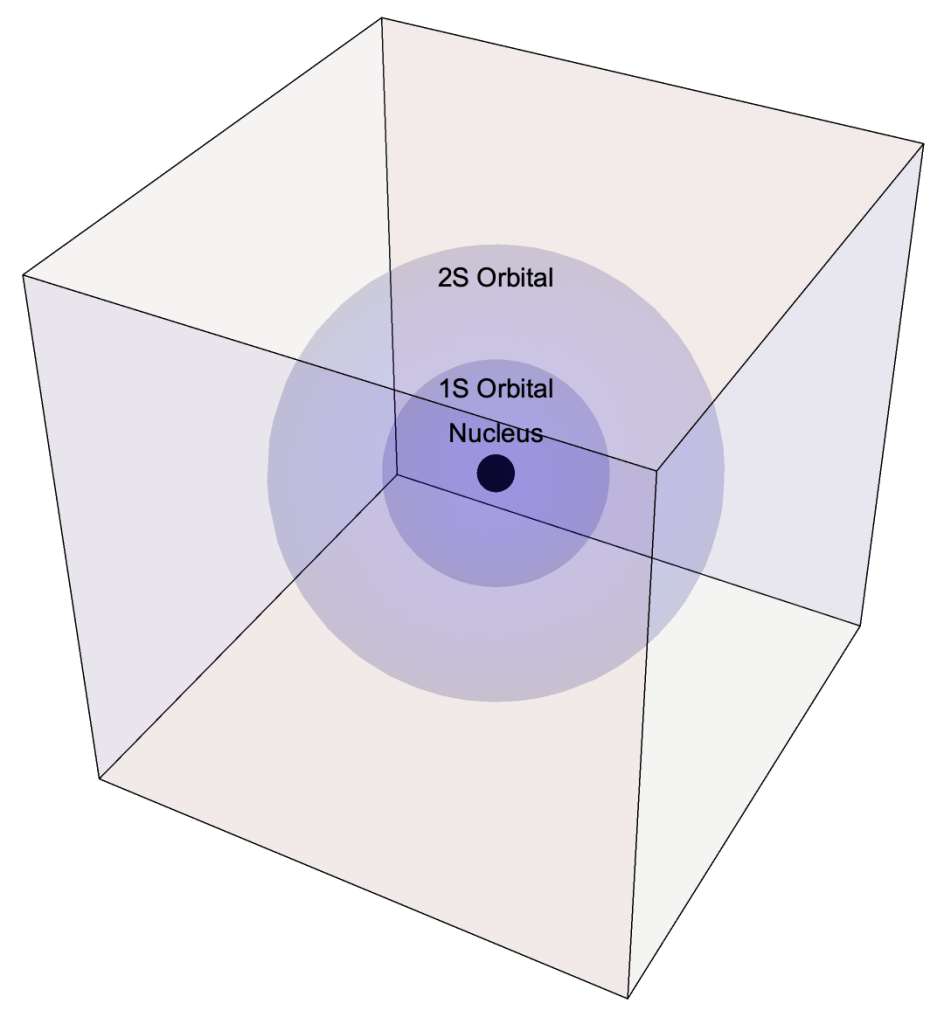
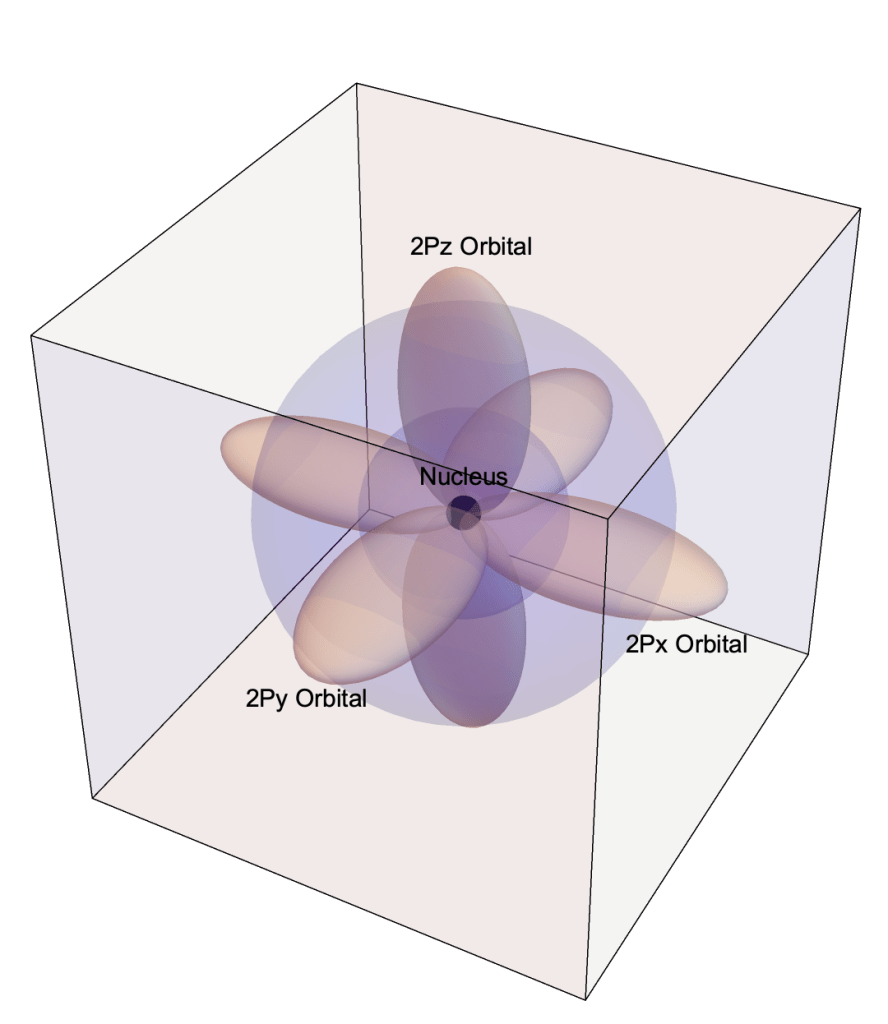
- The Quantum Atomic Model is derived from the Schrödinger Equation of Quantum Mechanics for the electric potential energy of system.
- In the Quantum Model electrons occupy orbitals rather than travel in orbits.
- An orbital is a three-dimensional region within which an electron is most likely observed.
- Orbitals are designated by a numeral, a letter, e.g. 1s, 2s, 2p, 3d, and an optional orientation specification.
- Numerals indicate an orbital’s energy level and relative distance from the nucleus.
- Letters s, p, d, f represent the orbital’s shape
- Orientations include: x, y, z, xy, yz, …
- Electrons and other subatomic particles have a property called spin, which is either Up or Down. The Pauli Exclusion Principle prohibits two electrons with the same spin from occupying the same orbital.
1s, 2s, 2px, 2py, and 2pz Orbitals
View Interactive Viewing Atomic Orbitals
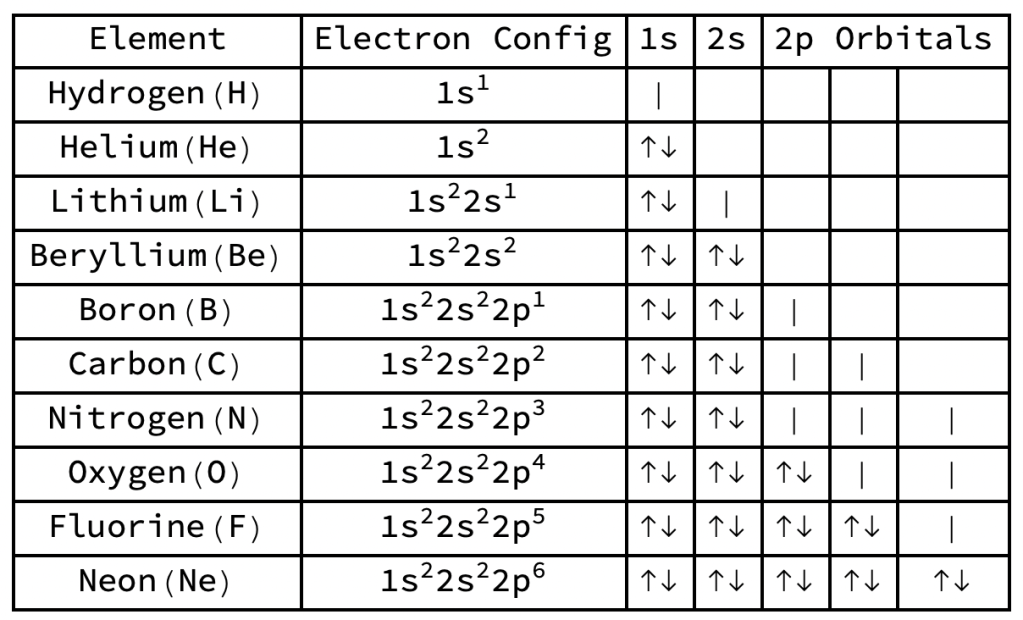
How electrons of first ten elements fill orbitals
- | indicates an electron whose spin may be Up or Down
- ↑ indicates an electron with spin Up
- ↓ indicates an electron with spin Down
- Orbitals shown are 1s, 2s, and the three 2p orbitals (2px, 2py, and 2pz)
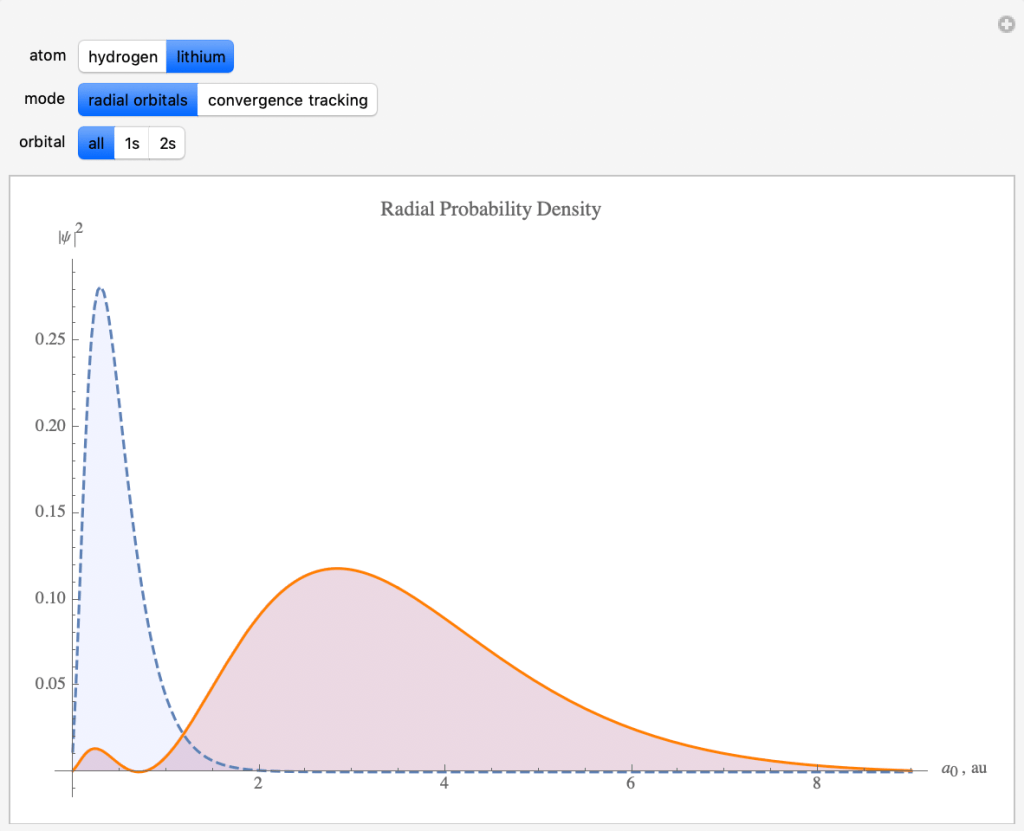
Hartree-Fock Method and Radial Probability Density
- britannica.com/science/quantum-mechanics-physics
- “The Schrödinger equation cannot be solved precisely for atoms with more than one electron, because of the number of particles and the variety of forces involved.
- Despite these difficulties, approximation methods introduced by the English physicist Douglas R. Hartree, the Russian physicist Vladimir Fock, and others in the 1920s and 1930s have achieved considerable success.”
- The Hartree-Fock Method simplifies calculations by using average locations and energies.
- The HF method was used to make this graph for the lithium atom, showing how likely lithium’s three electrons would be found at distances from the nucleus.
- There’s a high that the two electrons in the blue 1s orbital will be observed close to the nucleus.
- The electron in the red 2s orbital will be observed further away with greater uncertainty,
Periodic Table and Trends
Periodic Table
- Certain properties of the elements are periodic: as you run through the atoms by atomic number, you periodically encounter atoms with similar properties. The properties thus have trends.
- This is the basis for the Periodic Table of Elements
- The periodic properties are derived from Quantum Mechanics using approximate methods.
Periodic Trends, Examples
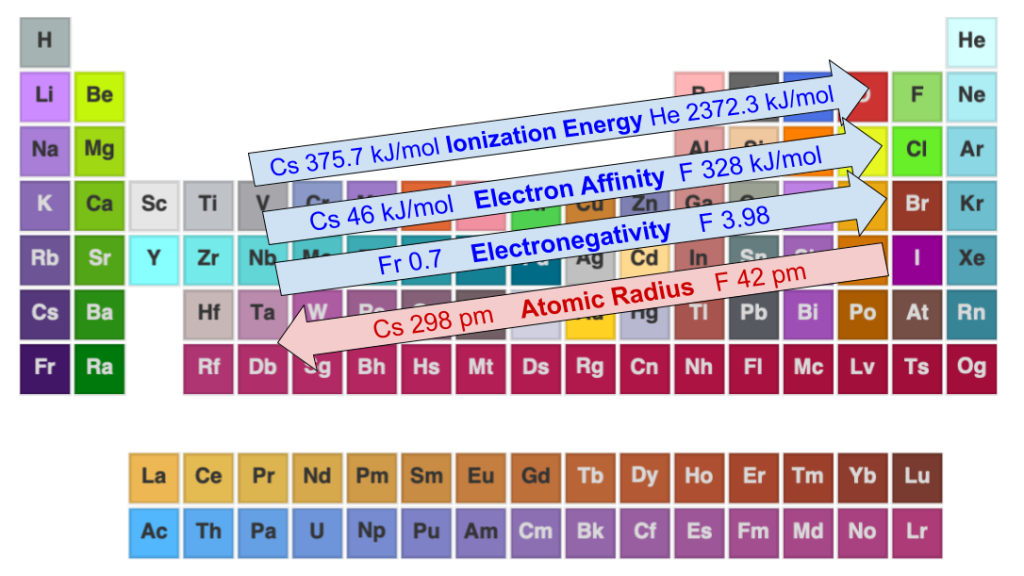
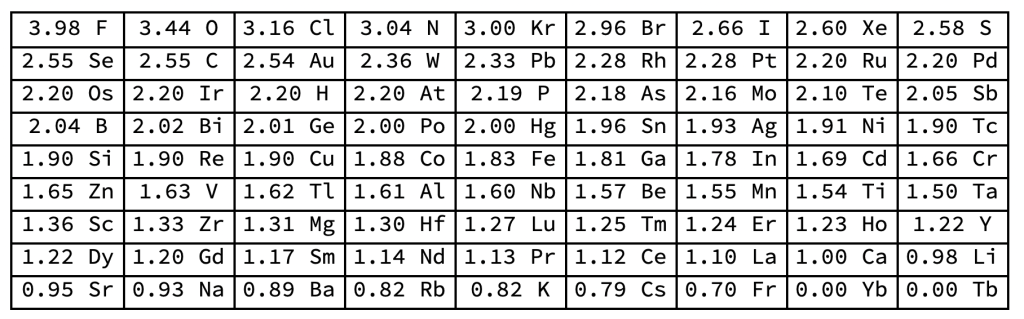
- Electronegativity is measure of how strongly an atom attracts an electron of another atom to be shared in a chemical bond. Values range from 3.98 (Fluorine) to 0.7 (Francium) on the Pauline Scale.
- Ionization Energy is the amount of energy required to remove an electron from an atom or molecule. For example, it takes 2372.3 kJ/mol to remove Helium’s outermost electron.
- Electron Affinity is the amount of energy liberated when an electron is added to a neutral atom to form a negatively charged ion. For example, adding an electron to an Oxygen atom releases 141 kJ/mol of energy.
- The Atomic Radius of an atom is “half the distance between the nuclei of identical neighboring atoms in the solid form of an element.” (Britannica)
Chemical Bonds
Overview
- A chemical bond is an electrostatic interaction among atoms or molecules, binding them.
- Chemical bonding is a matter of degree, depending on the strength of the electrostatic forces.
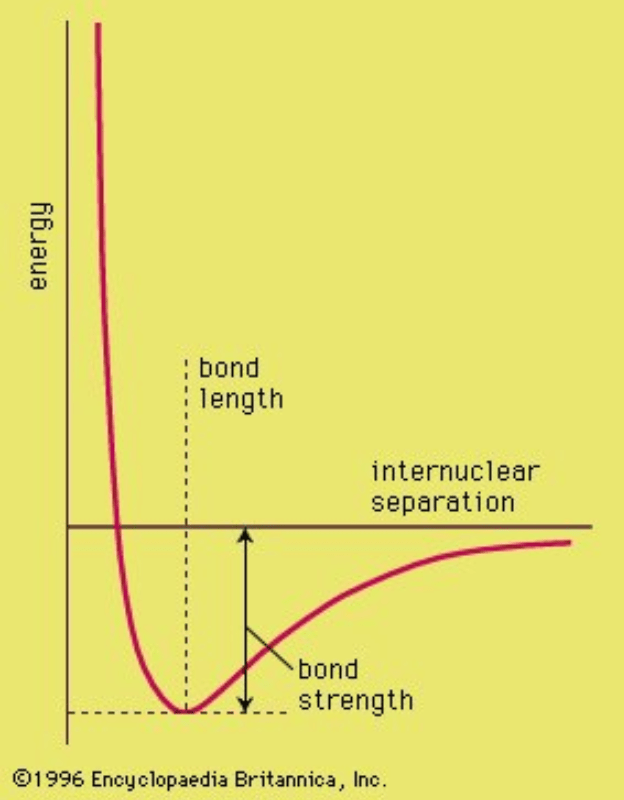
- The simplest bond is between two hydrogen atoms, creating an H2 molecule.

- Electrostatic attraction and repulsion of the two protons and two electrons of H2 determine the distance between the nuclei, represented by a plot of molecular potential energy against the internuclear separation. The bond length is where the potential energy is least.
Bonds among Atoms
The key to understanding bonds among atoms is electronegativity, the measure of how strongly an atom attracts an electron of another atom to be shared in a chemical bond. Values range from 0.7 (Francium) to 3.98 (Fluorine) to on the Pauline Scale.

Ionic Bonds
- An ionic bond is an interaction among atoms, resulting in a latticework of oppositely charged ions.
- The classic example is sodium chloride, NaCl, common table salt.
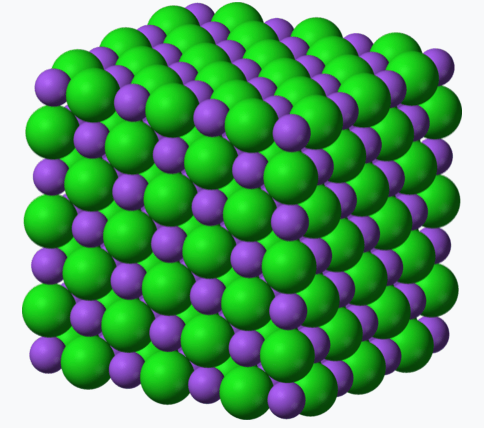
- The green spheres are positive sodium ions, Na+. The purple spheres are the smaller, negative chlorine ions, Cl–.
- The electronegativity of chlorine (3.16) is so much greater than sodium’s (0.93) that it steals an electron from the latter’s 3s shell and adds it to its p shell.
- Electron configurations of Na and Cl:
- Na = 1s22s22p63s1
- Cl = 1s22s22p63s23p5
- Electron configurations of Na and Cl:
- The result is an array of Na+ and Cl– ions.
- Two elements are unlikely to form an ionic compound unless one is a metal, in this case sodium.
Covalent Bonds
- A covalent bond is an interaction among atoms, resulting in electrons shared in molecule.
- Carbon dioxide is a molecule of one carbon and two oxygen atoms, sharing four electrons.
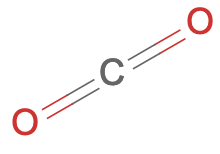
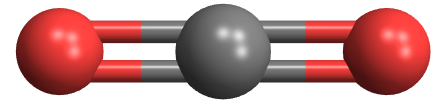

- Where Na and Cl form an ionic compound, carbon and oxygen become a molecule. The difference is the differences between their electronegativites.
- The difference between oxygen (3.44) and carbon (2.55) is 0.89, compared to 2.23 for Cl and Na. Thus oxygen’s electrostatic attraction for electrons is not strong enough to steal electrons from carbon. It’s forced to share. CO2 thus develops covalent bonds with two oxygen atoms, sharing two electrons with each.
- A molecule of water has one oxygen atom and two hydrogens, sharing two electrons.

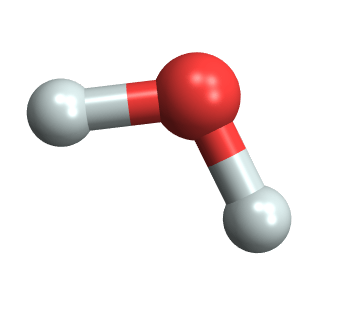

- Since oxygen’s electronegativity (3.44) doesn’t overwhelm hydrogen’s (2.20), O and H form covalent bonds, sharing two electrons.
- Electron configurations of O and H:
- O = 1s2 2s2 2p4
- H = 1s1
- Electron configurations of O and H:
- The water molecule is interesting because it’s bent, thanks to the shared electrons occupying oxygen’s p orbitals, which are oriented differently in space. Since oxygen’s electronegativity is greater than hydrogen’s, their shared electrons are closer to the oxygen nuclei. This makes the oxygen side of H2O slightly negative and the hydrogen side slightly positive. A water molecule is thus an electric dipole and its bonds polar covalent bonds.
- An electric dipole is a pair of equal and opposite electric charges, physically separated.
- Dipole moment is a measure of a molecule’s dipole strength.
- The dipole moment of H2O is 1.8546 debyes. That of CO2 is N/A.
Metallic Bonds
- A metallic bond is an interaction among metal atoms, resulting in a latticework of positive metal ions in a “sea of electrons.”
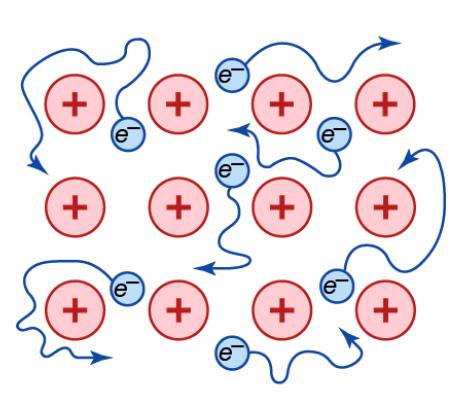
- Simple metals like aluminum, magnesium, and sodium lose their valence (outer) electrons in a “sea of electrons.”
- Transition metals like iron, copper, and titanium lose some of their valence electrons in the sea. Other electrons move from ion to ion through shared covalent bonds of electrons in d-orbitals.
- Transition metals such as iron can form compounds with small atoms filling in the holes of the metallic latticework. In Iron Carbide, Fe3C, carbon atoms fill the holes.
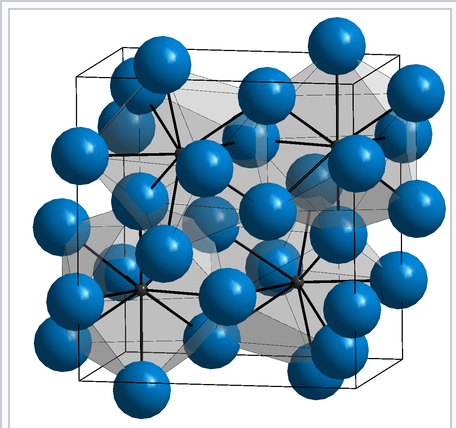
Image Credit: wikipedia.org/wiki/Cementite
Bonds among Molecules (Molecular Bonds, Van der Waals Forces, Intermolecular Forces)
- An intermolecular bond is an electrostatic interaction among molecules.
- The basis for the interaction is the electric dipole, a pair of equal and opposite electric charges, physically separated.
- Intermolecular bonds are of three kinds:
- Polar
- Induced Polarity
- London (Dispersion) Forces
Polar Bonds
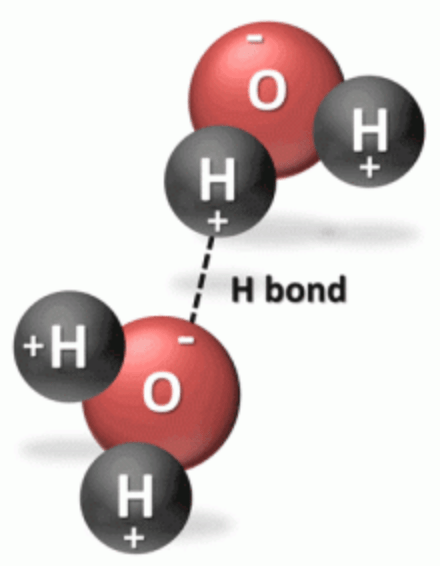
- A polar intermolecular bond is an interaction among polar molecules. Being polarized, the oxygen atom of one H2O molecule is attracted to a hydrogen molecule of another.
Induced Polarity Bonds
- An induced polarity bond is an interaction in which a polar molecule distorts the electron charge of a nearby non-polar molecule or atom, inducing temporary polarity.
- Here HCl, a polar molecule with H having a slight positive charge, disrupts the electrons of an Argon atom.


London (Dispersion) Forces
- Electron fluctuations in molecules cause instantaneous dipoles, which can induce brief polarization in nearby molecules.
Chemical Reactions
- A chemical reaction is the transformation of substances (the reactants) into different substances (the products) by the rearrangement of atoms.
- Chemical reactions are distinct from physical changes, such ice melting to water and water evaporating to steam. In a physical change a substance changes properties but retains its identity.
- Chemical reactions are also distinct from nuclear reactions, such as nuclear fusion and fission, which involve the transformation of atomic nuclei.
- Chemical reactions are governed by:
- the Electrostatic Force per Quantum Mechanics
- the First and Second Laws of Thermodynamics
- the Conservation of Mass for Chemical Reactions
- Examples of Reactions
Combustion of Methane
- Turn a burner on, the gas ignites, and there’s a flame.

- The gas is natural gas, a mixture of mostly methane and ethane.
- I’ll talk just about methane.
- The combustion of methane is a chemical reaction, methane and oxygen transformed into carbon dioxide and water vapor.
- The reaction is represented by equations and diagrams:
Chemical Equation
CH4(g) + 2O2(g) → CO2(g) + 2H2O(g)
Stick and Ball Diagrams
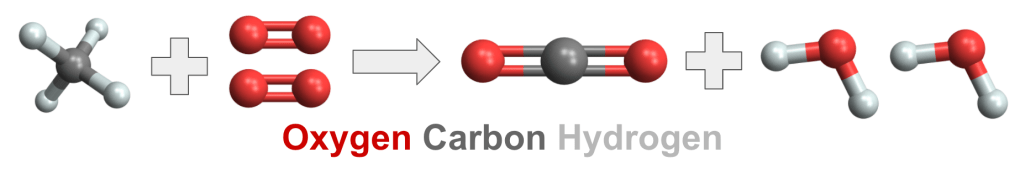
Structural Formulas
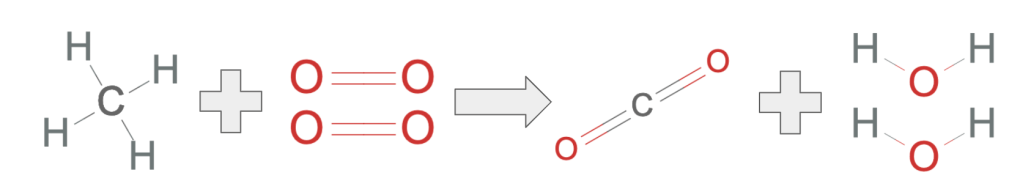
- In the reaction, the atoms rearrange themselves. The 4 hydrogens bonded with the carbon atom attach themselves to two of the four oxygens. The other oxygens bond with the carbon.
- But why?
- Why do the O, H, and C atoms rearrange themselves into CO2(g) + 2H2O(g) rather than remain CH4(g) + 2O2(g)?
First, a necessary condition must be satisfied
- The Conservation of Mass for Chemical Reactions requires that the total mass of the particles during methane’s combustion remain the same. Such is the case:
- Reactant Side
- Mass of CH4 = 16.043 g/mol
- Mass of O2 = 31.998 g/mol
- Total = 16.043 + 2(31.998) = 80.039 g/mol
- Product Side
- Mass of CO2 = 44.009 g/mol
- Mass of H2O = 18.015 g/mol
- Total = 44.009 + 2(18.015) = 80.039 g/mol
- Reactant Side
- The Conservation of Mass requires that chemical equations are balanced, that is, the Reactant and Product Sides must have the same number of atoms of each element, in this case one carbon, four oxygens, and four hydrogens.
Intuitive Explanation
- Atoms rearrange themselves to form CO2(g) + 2H2O(g), rather than remain CH4(g) + 2O2(g), because oxygen has a stronger electrostatic attraction for nearby electrons than carbon.
- Two important concepts:
- Electronegativity is measure of how strongly an atom attracts an electron of another atom to be shared in a chemical bond. Values range from 3.98 (Fluorine) to 0.7 (Francium) on the Pauline Scale.
- Valence is the number of covalent bonds an atom typically forms
- Pertinent values:
- Oxygen
- Electronegativity = 3.44
- Valence = 2
- Hydrogen
- Electronegativity = 2.20
- Valence = 1
- Carbon
- Electronegativity = 2.55
- Valence = 4
- Oxygen
- Because oxygen’s electronegativity is larger than carbon’s, two of the four oxygens form covalent bonds with the four hydrogens to make water vapor. This leaves the other two oxygens to form covalent bonds with carbon.
Explanation in terms of Gibbs Free Energy
- In the 1870s Josiah Willard Gibbs developed the ingenious concept of Gibbs Free Energy, which combines internal energy (from the First Law of Thermodynamics) with entropy (from the Second Law).
- Here’s the procedure:
- Look up the Gibbs Free Energies of Formation (ΔGf°) for the molecules in the combustion of methane in the Table of Standard Gibbs Free Energies.
- ΔGf° of CO2 = −394.39 kJ/mol
- ΔGf° of O2 = 0 kJ/mol
- ΔGf° of H20 = −237.14 kJ/mol
- ΔGf° of CH4 = -50.5 kJ/mol
- Calculate the change ΔG in Gibbs Energy for the reaction by subtracting the Gibbs Energies for the reactants from the Energies for the products.
- Reactants
- CH4(g) + 2O2(g) = -50.5 + 2 * 0 = -50.5
- Products
- CO2(g) + 2H2O(g) = −394.39 + 2 * (−237.14) = -868.67
- ΔG = -868.67 – (-50.5) = -818.17
- Reactants
- Look up the Gibbs Free Energies of Formation (ΔGf°) for the molecules in the combustion of methane in the Table of Standard Gibbs Free Energies.
- The fact that ΔG is negative means that the combustion of methane is spontaneous and exoergic.
- A spontaneous process is one that, once started, requires no external energy.
- A chemical reaction is exoergic (ex-oh-ERR-jick) if it releases energy.
- So the combustion of methane, once started, keeps going (as long as there’s methane and oxygen). The reaction also releases energy in the form of heat:
- CH4(g) + 2O2(g) → CO2(g) + 2H2O(g) + Energy
View Gibbs Free Energy
Heat Produced by the Combustion
- The amount of heat produced by the combustion of methane can be calculated using the concept of enthalpy (EN-THOWL-pea).
- Look up the standard enthalpies of formation for the molecules in the Table of Standard Enthalpies.
- ΔH for CO2 (gas) = −393.509
- ΔH for O2 (gas) = 0
- All elements have zero enthalpies
- ΔH for H2O (liquid) = −285.8
- ΔH for CH4 (gas) = −74.9
- Compute ΔH for the reaction by subtracting the enthalpies for the reactants from the enthalpies from the products:
- Reactants
- CH4(g) + 2O2(g) = (−74.9) + 2 * 0 = -74.9
- Products
- CO2(g) + 2H2O(g) = (−393.509) + 2 * −285.8 = -965.109
- ΔH = -965.109 – (-74.9) = -890.209 kJ/mol
- Reactants
- The combustion of methane releases 890.209 kJ/mol of heat.
View Enthalpy
Photosynthesis
- Plants use photosynthesis to convert carbon dioxide and water to glucose.
- Chemical Reaction
- 6 CO2 (g) + 6 H2O (l) → C6H12O6 (s) + 6 O2 (g)
- 6 carbon dioxides + 6 waters → 1 glucose + 6 oxygens
- Gibbs Free Energy
- Gibbs Energies for the molecules
- ΔGf° of CO2 = CO2 = -394 kJ/Mol
- ΔGf° of Water = -237.14 kJ/Mol
- ΔGf° of Glucose = -910.56 kJ/Mol
- ΔGf° of O2 = 0 kJ/Mol
- ΔG = (-910.56 + 6*0) – (6 * -394 + 6 *-237.14) = +2876.28 kJ/Mol
- The fact that ΔG is positive means that photosynthesis is non-spontaneous process and endoergic
- A non-spontaneous process requires constant external energy to keep going.
- A chemical reaction is endoergic (end-oh-ERR-jick) if it absorbs energy.
- Photosynthesis uses radiant energy to keep the process going.
- 6 CO2 (g) + 6 H2O (l) + Energy → C6H12O6 (s) + 6 O2 (g)
- Gibbs Energies for the molecules
View Gibbs Free Energy
- Enthalpy can be used to determine the amount of energy photosynthesis requires.
- Enthalpies for the molecules
- ΔH of CO2 = -393.513 kJ/mol
- ΔH of Water = -285.84 kJ/mol
- ΔH of Glucose = −1275.0 kJ/mol
- ΔH of O2 = 0 kJ/mol
- ΔH = (−1275.0) – ( 6 *-393.513 + 6 * -285.84) = +2801.12 kJ/mol
- So photosynthesis requires 2801.12 kJ/mol of energy to keep the reaction going.
- Enthalpies for the molecules
View Enthalpy
Carbonation
- The change in Gibbs free energy for the combustion of methane is negative, signifying that the reaction is spontaneous and releases energy.
- For photosynthesis the change is positive, meaning the reaction is non-spontaneous and requires energy.
- For carbonation, the process of making beverages bubbly by adding carbon dioxide, the change in Gibbs free energy is virtually zero, meaning that the system is in chemical equilibrium.
- The reaction is:
- H2CO3(aq) ⇌ CO2(g)+H2O(l)
- 1 carbonic acid is in chemical equilibrium with 1 carbon dioxide + 1 water.
- Gibbs Energy
- The molecules
- ΔG for CO2 = -394 kJ/Mol
- ΔG for Water = -237.14
- ΔG for Carbonic acid = −623.1
- ΔG = (-394 + -237.14) – −623.1 = -8.04
- The molecules
- When carbon dioxide is dissolved in water, a small portion is converted to carbonic acid, the rest remaining as CO2. Carbonic acid, however, spontaneously decomposes to carbon dioxide and water. Thus, opposite reactions occur simultaneously, producing a dynamic equilibrium.
Addendum
Chemical Equations
- A chemical equation is a top-level representation of a chemical reaction that
- has the form:
- Reactant-Side ………. Arrow ……….. Product-Side
- consists of chemical formulas, numerals, arrows, and the symbols: +, (g), (l), (s) (aq)
- has the form:
- Example:
- CH4(g) + 2O2(g) → CO2(g) + 2H2O(g)
- The Reactant Side represents the reactants present at the beginning of the reaction.
- The Product Side represents the products present at the end of the reaction.
- Arrows
- → is an irreversible reaction,
- ⇄ is a reversible reaction
- ⇋ is a reversible reaction in equilibrium
- For other arrows:
- (g), (l), (s), (aq) indicate gas, liquid, solid, aqueous solution
- The Conservation of Mass requires that a chemical equation is balanced, that the Reactant and Product Sides have the same number of atoms of each element.
Enthalpy and Gibbs Free Energy
View Enthalpy and Gibbs Free Energy
Molecular Orbital Theory
- Molecular Orbital Theory, developed by Robert Mulliken and Friedrich Hund in 1927, is the main quantum mechanical theory of molecular structure. The basic idea is to treat a molecule as a big atom with electrons occupying molecular orbitals instead of atomic orbitals.
- We’ll use MOT to answer the question:
- Why do two hydrogen atoms combine to form the molecule H2 but two helium atoms don’t form He2?
- That is, why is it that 2H → H2 but not 2He → He2?
- Procedure: Try to build a molecule from atoms:
- Create molecular orbitals for the molecule, building them by adding and subtracting the atomic orbitals.
- Move the nuclei of the atoms to the center of each molecular orbital.
- Move the electrons one by one from the atoms to the molecular orbits with the lowest energy.
- When the molecule is complete see if its energy is less than the sum of the energies of the original atoms. If so, the atoms in the real world will combine to form a molecule. If not, real atoms won’t combine.
- Let’s try forming an H2 molecule from two hydrogen atoms. The atoms are at the left and right of the diagram, each having a single electron in the 1s orbital.
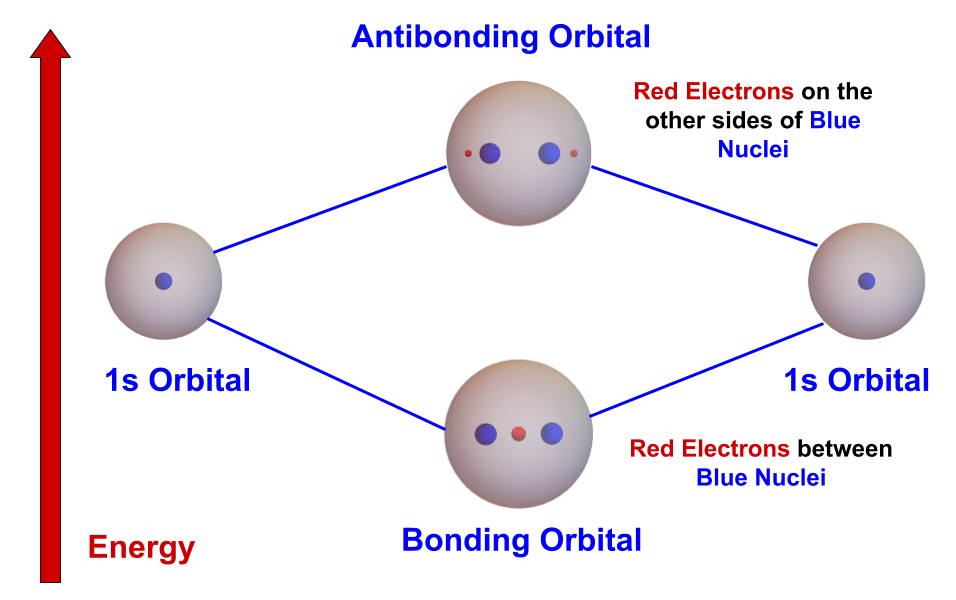
- First we build the molecular orbitals of the molecule from the 1s atomic orbitals. By adding the atomic orbitals we create the lower-energy, spherical bonding orbital. By subtracting the atomic orbitals we create the higher-energy, two-sphere antibonding orbital. We then put the nuclei next to each other in the two molecular orbitals.
- Now we move the electrons. There are two rules:
- Move an electron to the molecular orbital with the lowest energy.
- Each orbital can hold only two electrons.
- So both hydrogen electrons end up in the lower-energy bonding orbital. Since there are no electrons in the higher-energy antibonding orbital, the total energy of H2 is less than the energies of the hydrogen atoms. Thus H2 forms.

- Now let’s try forming an He2 molecule from two helium atoms. The procedure is the same as for hydrogen except there are four electrons to be moved. The same rules apply:
- Move the electron to the molecular orbital with the lowest energy.
- Each orbital can hold only two electrons.
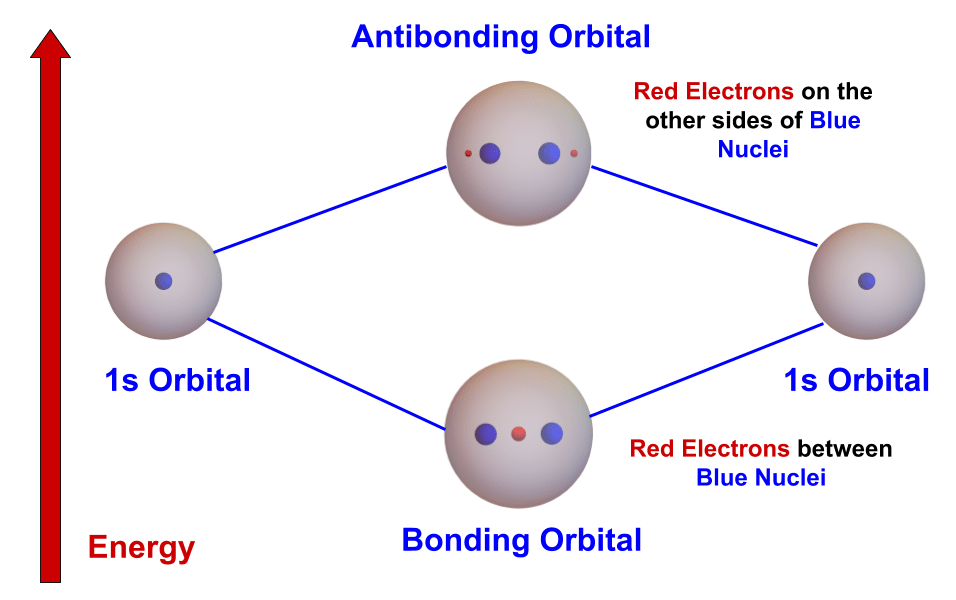
- The first two electrons are moved to the lower-energy bonding orbital. The other two electrons, however, must be moved to the higher-energy antibonding orbital, since the bonding orbital is full.
- So the four electrons are evenly divided between the lower and higher energy orbitals. The sum of the energies is no greater than the sum of the helium atoms. Therefore no He2 molecule forms.

View Interactive Molecular Orbital Theory for Possible Diatomic Molecules